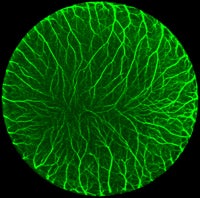
Research in my laboratory is focused on elucidating the cellular and molecular mechanisms involved in ocular development and embryonic corneal wound healing. The cornea is a highly specialized transparent tissue located at the anterior-most surface of the eye. Multipotent cells known as the neural crest give rise to majority of the cells in the cornea including the stromal keratocytes, corneal endothelium, and sensory nerves. Development and wound healing of the cornea are both multi-step processes that involve coordinated migration and differentiation of cells, as well as the intricate patterning of sensory nerves and ocular blood vessels. Using avian and mice as a model organisms, combined with molecular, bioinformatics, microsurgical, and tissue/cell culture approaches, we have shown that: 1) Periocular neural crest cell migration and differentiation are tightly regulated processes during corneal development. 2) Corneal keratocytes retain the stem cell-like properties of their neural crest progenitors. 3) Semaphorins and chemokines regulate sensory innervation of the cornea, neural crest cell migration, and vascular patterning in the anterior eye. Our ongoing research is aimed at further elucidating the mechanisms involved in periocular neural crest cell differentiation and the role of the extracellular matrix during this process. Since corneal regeneration recapitulates development, we extrapolate our studies to cornea wound healing. We have recently begun to study the stem cell potential of periocular neural crest cells in the embryonic corneal environment. Ultimately, the goal of our research is to understand how signaling between the ocular tissues and corneal cells is disrupted in congenital eye disorders and corneal wound healing. Our findings may provide insights into new treatments for vision threatening corneal defects and injuries.

