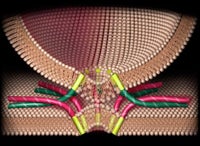
My lab is interested in the molecular mechanisms of membrane fusion. Membrane fusion is a fundamental process involved in diverse cellular events such as fertilization and neuroscretion. Biological membrane fusion relies on proteins to drive membrane merger. A family of integral membrane proteins collectively known as SNAREs mediates the fusion of intracellular transport vesicles. While SNAREs pair in specific ways to provide the mechanical energy to drive fusion, the delicate interplay of regulatory elements that orchestrate this event in space and time remain elusive. We use biochemistry, cell biology and molecular genetic techniques to study the assembly and regulation of SNARE proteins in two model systems: vesicular transport in the budding yeast S. cerevisiae and neurosecretion in the fruity D. melanogaster. We have initially focused our attention on the regulatory role of Sec1p in yeast exocytosis. We have recently shown that Sec1p binds primarily to the yeast t-SNARE complex and directly stimulates membrane fusion. We are currently examining the precise method of Sec1p stimulation as well as analyzing the function of the Sec1p family member ROP in neuronal exocytosis. Additional studies in the lab involve membrane fusion events in other aspects of yeast cell biology including sporulation and cell-cell fusion during mating.

