Research Summary
Dr. Billups' early research interests included the synthesis of new molecular systems of theoretical interest. A major focus involved the development of a vacuum gas phase procedure for the synthesis of small ring cycloalkenes using reagents adsorbed on inert surfaces to affect elimination reactions (figure 1). This approach allows the reactive species to be isolated at low temperature, and eliminates many of the undesired bimolecular side reactions that would normally be encountered in solution. Energetic compounds can thus be collected readily in cold traps for further chemical studies or spectral characterization. X-ray structural parameters of unstable low melting compounds were secured in collaboration with Dr. Roland Boese in Essen, Germany. Representative molecules whose syntheses have been accomplished include methylenecyclopropene, spiropentadiene, and spiroheptatriene. 3,3’-Bicyclopropenyl, the last (CH6) isomer benzene, was first made only in 1989 and characterized by X-Ray crystallography in the Boese laboratory. Collaborative work with Boese and an industrial sponsor led to the nucleation, growth, and structure of gas hydrates by X-Ray crystallography. Gas hydrates cause serious problems in long distance natural gas pipelines.
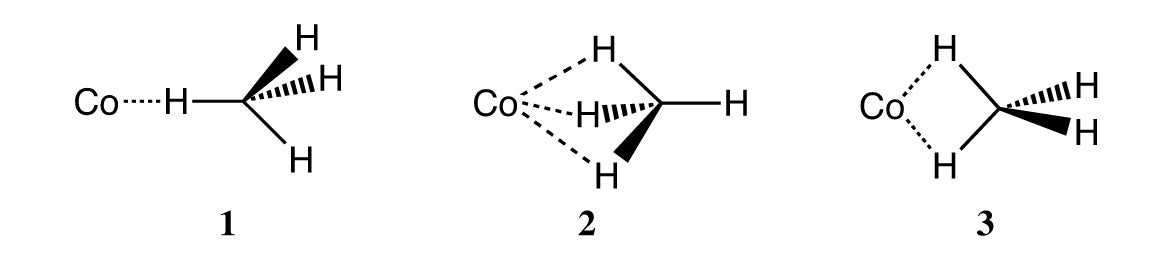
A second area involved studies on the activation of carbon-hydrogen bonds by first row transition metals (figure 2). For example, photoexcited cobalt atoms were found to insert into the carbon-hydrogen bonds of methane to yield CH3CoH. This same species can be microsynthesized and characterized using FTIR spectroscopy by cocondensing the metal with CH2N2, H2, and argon at 11 K. Photolysis of CH3CoH using 400 nm light leads to extrusion of the metal with the formation of the spectroscopically detectable Co(CH4) σ-complex 2 shown to have C3v symmetry. Photolysis of this complex using a UV source regenerates the insertion product CH3CoH. Deuterium labeling studies have shown that the methane rotates freely on the cobalt. These studies were carried out in collaboration with the late Professor John Margrave.
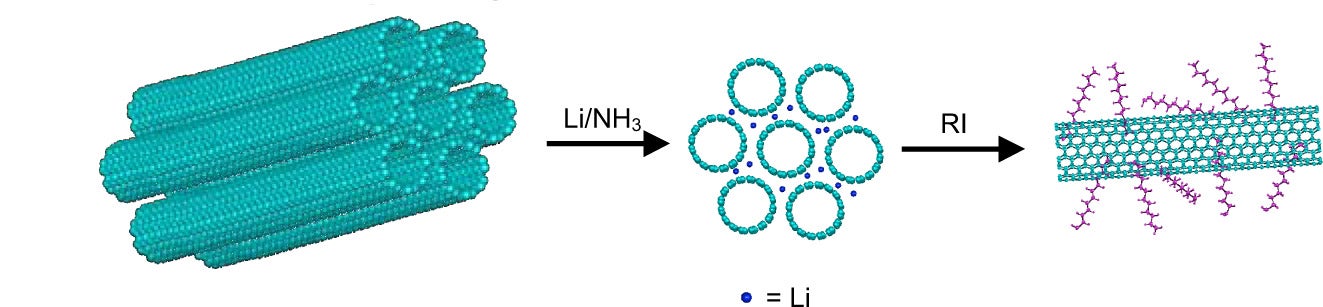
Current studies in the area of carbon nanomaterials have focused on the development of routes to soluble carbon nanotubes and graphene (figure 3). Reductive alkylation reactions that give nearly complete exfoliation of these materials have been developed. Application of these reactions to coal has allowed us to synthesize soluble coal nanoplatelets (liquid coal).